
In the case of period 2, there are two core electrons in the #"1s"# subshell.įor the transition metals, determining valence electrons is tricky because they can use inner electrons as valence electrons. Valence is the number of connections an atom tends to form. The rest of the electrons are the core electrons. Scientists soon observed patterns in the valence of the different elements. 10":#"1s"^2"2s"^2"2p"^6"#įor all of the preceding elements, the valence electrons are the outermost (highest energy) #"s"# and #"p"# electrons. A neutral atom has the same number of electrons as protons. The atomic number is the number of protons in the nuclei of the atoms of an element. The electron configuration shows that the last shell of krypton has. 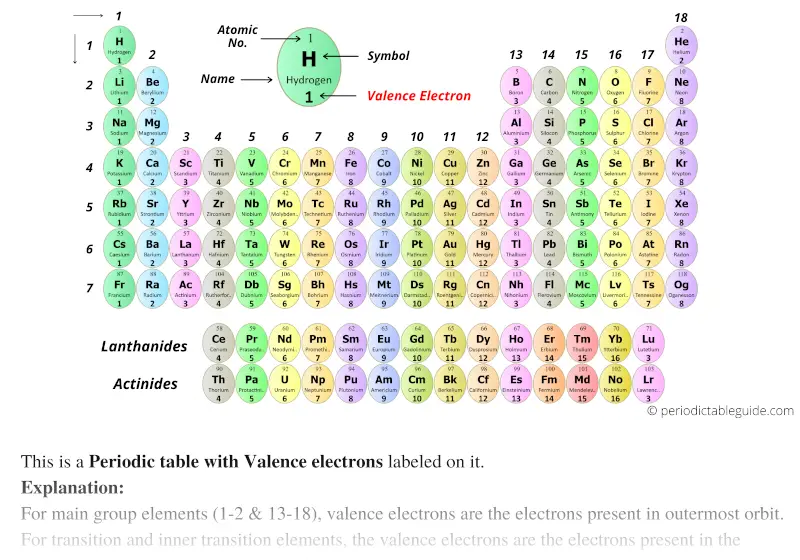
The total number of electrons in a valence shell is called valence electrons. In single covalent bonds, typically both atoms in the bond. Valence electrons are outer shell electrons with an atom and can participate in the formation of chemical bonds. The last shell after the electron configuration is called the valence shell. This page titled 4: Valence Electrons and Bonding is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by Elizabeth Gordon. And so, if I look at period 1, and I just move across my periodic table, hydrogen is in the first period and so is helium. A period is a horizontal row on the periodic table. And so lets move on to the concept of periods. The third step is to diagnose the valence shell. And this second way of numbering your groups is useful when youre thinking about valence electrons. You can also find the core and valence electrons by determining or looking up the electron configurations of the main group elements. Step-3: Determine the valence shell and calculate the total electrons. Across a period, elements in group 1/IA have one valence electron, elements in group 2/IIA have two valence electrons, elements in group 13/IIIA have three valence electrons, and so on, ending with group 18/VIIIA, which have eight valence electrons, which is the maximum number of valence electrons. You can determine the number of valence electrons in the atoms of the main group elements by the group number of the element. Using Cannizzaros atomic weights, Mendeleev and Lothar Meyer made a great discovery, the periodic law: If. aluminum oxide, Al 2 O 3: aluminum has a valence of 3. hydrogen sulfide, H 2 S: sulfur has a valence of 2. The core electrons are in the inner shells and do not participate in chemical reactions. For main group elements, the number of valence electrons usually ranges between 1 and 8 because eight electrons forms a complete octet. Valence electrons are the electrons present in the outermost shell of an atom. lithium oxide, Li 2 O: lithium has a valence of 1. The main group elements are the A groups, or groups 1,2,13-18. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. The valence electrons participate in chemical reactions. Thus, the periodic table becomes a tool for remembering the charges on many ions. Lithium has a single electron in the second principal. In the second period elements, the two electrons in the 1 s sublevel are called inner-shell electrons and are not involved directly in the elements reactivity, or in the formation of compounds. \) can both react with water, but K has a more radical reaction because it has more shells of core electrons which makes the valence electron in its outermost orbital much easier to lose than the valence electron of Na.For the main group (representative) elements, the valence electrons are the outermost (highest energy) #"s"# and #"p"# electrons, which make up the valence shell. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. The paper introduces Chemdex, a freely accessible web-based database of over 70 000 compounds characterised by crystallography from across the periodic table. Valence electrons are the electrons in the highest occupied principal energy level of an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
